Many diseases occur when microbes from the environment invade our body. Others arise when the immune system goes rogue and attacks the body from within. “It is unable to judge cells that belong to our body from anything else that comes from the outside. This is when autoimmune disease occurs,” explains Dr Savita Devi, a faculty in the Department of Biotechnology, who studies immunity and inflammation.
Scientists, Prof Mary E Brunkow from the Institute for Systems Biology, Seattle, Prof Fred Ramsdell from Sonoma Biotherapeutics, San Francisco, and Prof Shimon Sakaguchi from Osaka University, discovered a new safety mechanism that protects the body from such self-inflicted damage. Their findings showed that chinks in this system lead to autoimmune disease, opening potential avenues of therapy. The scientists won the 2025 Nobel Prize in Physiology or Medicine.
Recognising self
There are two arms of the body’s immune system, a first line of defense that responds to threats more broadly, and a second that specifically recognises and targets pathogens. The Nobel Prize-winning research focused on this specialised arm, particularly immune cells called T cells.
For a long time, scientists knew of two types of T cells, one that detects microbes, and another that attacks virus-infected cells and tumour cells. These immune cells carry receptors on their surface that specifically recognise intruders based on their unique molecular signatures. However, T cells that recognise the body’s own cells and mount an attack against them are harmful.
“We have evolved a mechanism where immune cells carrying receptors that recognise our own cells or host structures are destroyed,” says Dr Savita. Soon after birth, such self-reactive T cells are swiftly eliminated in a gland called the thymus, which is present in our neck. This mechanism is known as ‘central tolerance’.
A safety back-up
Until the mid-90s, central tolerance was thought to be the only mechanism by which the body protects itself from autoimmunity. However, research by Brunkow, Ramsdell and Sakaguchi revealed a new class of cells, regulatory T cells, that dampens autoimmune responses. This safety back-up came to be known as ‘peripheral immune tolerance’. The clinching evidence came when the scientists discovered that certain mutations in a gene crucial for regulatory T cell development lead to severe autoimmune disease.
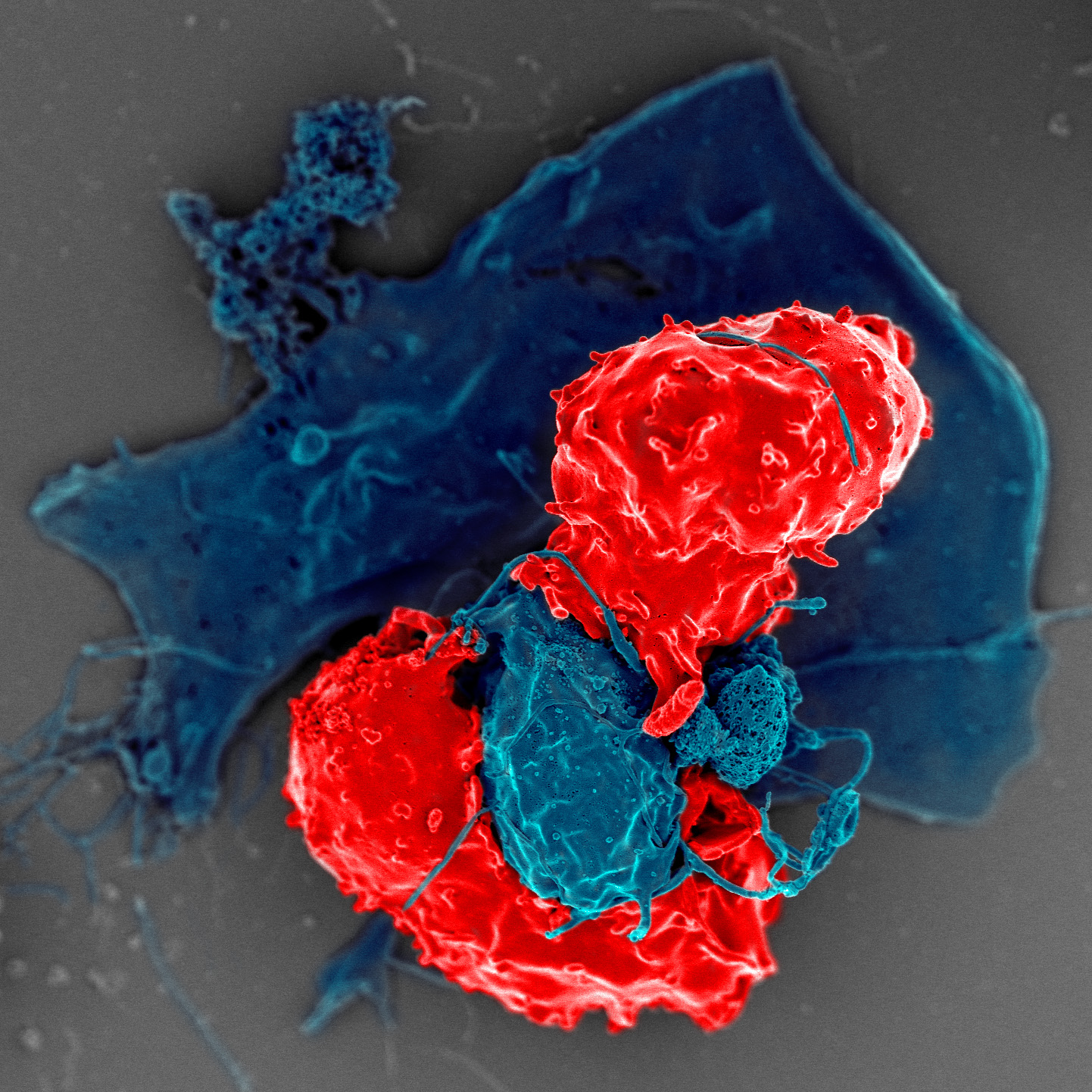
We now know that peripheral immune tolerance works by detecting errors made by central tolerance. When self-reactive T cells escape elimination in the thymus and start attacking the body, regulatory T cells intervene and de-escalate the reaction. They keep the immune system in check by secreting anti-inflammatory cytokines, which are proteins that regulate immunity.
So far, the story has focused on how tolerance of the body’s own cells is built into the specialised arm of the immune system, and how its failure leads to autoimmune disease. Dr Savita’s group is interested in exploring the other side of the coin: how chronic inflammation, driven by the activation of protein complexes called inflammasomes, leads to various diseases, such as infectious diseases, metabolic diseases, and cancer.
The other side of immunity

Inflammation receives a bad rap, but it is an important physiological process that helps the body respond to infection and injury. During inflammation, immune cells attack pathogens to eliminate them. But sometimes, invaders escape and enter the immune cell. When this happens, sensors inside the cell detect the intruder and trip an alarm system, which leads the cell to self-destruct to contain the infection.
A key component of the alarm system is a protein complex called the inflammasome, which triggers the release of specific cytokines that dial up inflammation. They induce the formation of holes in the cell membrane and eventually the cell gets destroyed. “It is a very inflammatory form of cell death,” says Dr Savita.
Her research group is interested in understanding how inflammasomes regulate inflammation in the body. By performing experiments on immune cells grown in the lab, they are trying to understand how the function of inflammasomes is regulated, and how imbalances lead to disease.
Recent studies have linked inflammasomes to a range of diseases, which opens novel avenues for therapy. The immune system’s dual lines of defence are also gaining attention in cancer research. Tumour cells may exploit regulatory T cells to escape immune attack and suppress inflammasome activation to escape cell death. Researchers are exploring ways to target both these escape mechanisms used by cancer cells. Uncovering the intricate mechanisms that keep the immune system in check can help scientists understand why diseases arise and how we can treat them.