Around ten years ago, when Dr Deepu Babu was studying carbon nanomaterials for his PhD, he saw a flurry of research on a new material that scientists were excited about. “It was probably the golden time for metal-organic frameworks (MOFs),” he says, now a faculty in the Department of Materials Science and Metallurgical Engineering. “That is when I decided to work on MOFs.”
The scientists who pioneered the development of this material – Prof Susumu Kitagawa from Kyoto University, Prof Richard Robson from the University of Melbourne, and Prof Omar M Yaghi from the University of California, Berkeley – won the 2025 Nobel Prize in Chemistry.
Materials that store and sieve
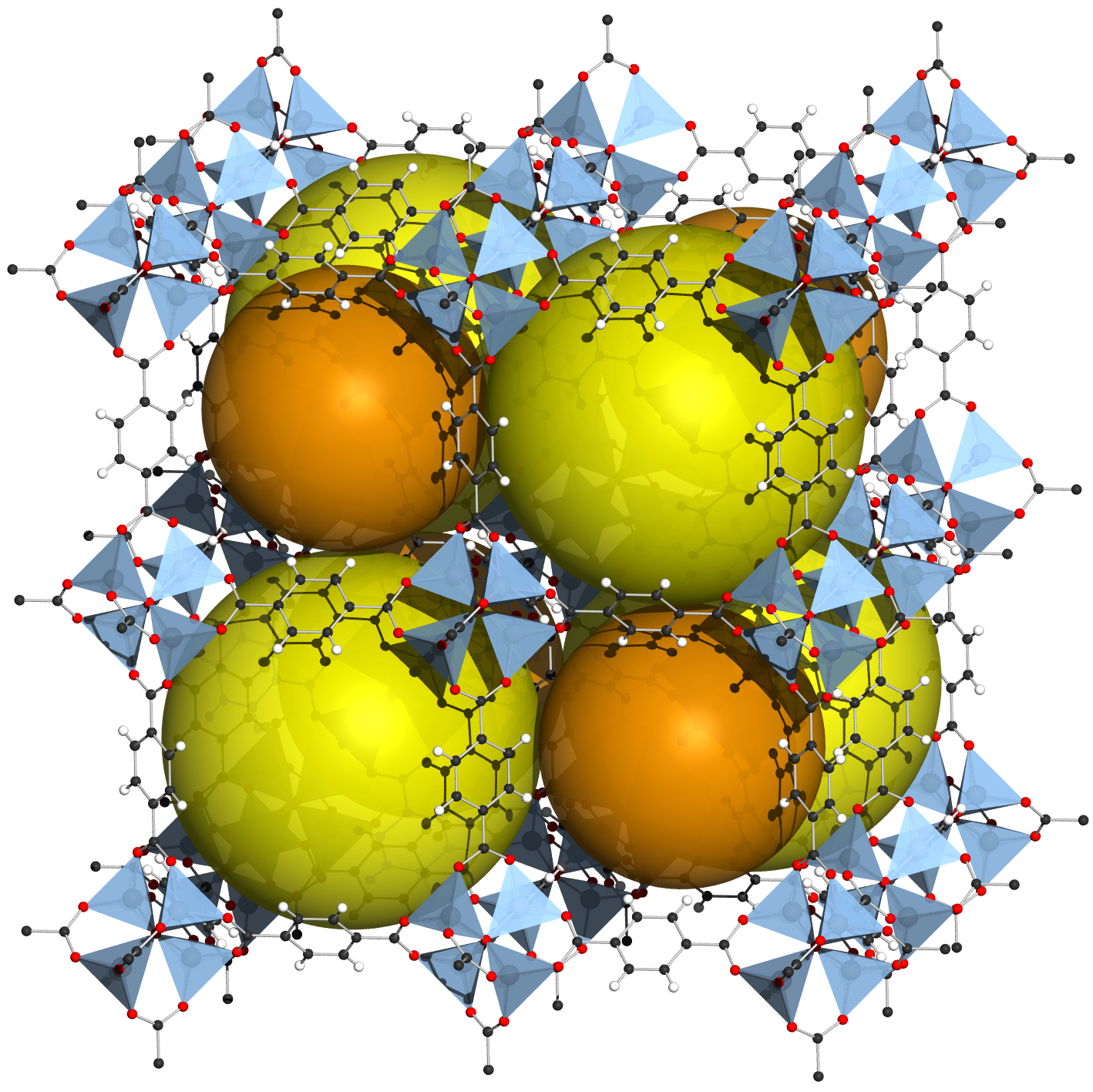
Metal-organic frameworks are materials where large organic molecules link metal ions to create a roomy scaffold where other molecules can sit or pass through. While the first of these materials was synthesized in the late eighties, it took a decade or more to develop stable forms that could be put to use. These materials act like magical sieves, selectively capturing molecules from a mixture, which can then be stored or extracted separately.
Separation processes are very energy-intensive, accounting for close to 15% of global energy consumption. This makes metal-organic frameworks valuable for various applications, such as separating deceptively similar chemicals, storing fuel, protecting people from toxic gases, and extracting water from desert air.
Space and stability
Surface area and pore size are two important characteristics of materials used in separation processes. Metal-organic frameworks have an internal surface area many times larger than conventional materials used in adsorption-based separation. Their ordered crystalline nature also creates well-defined pores, where the size of the pore is shaped by underlying molecules and their arrangement. A uniform pore architecture allows metal-organic frameworks to be used in challenging problems involving membrane-based separation.
Among the earliest stable materials developed in this class was MOF-5, which has a very large surface area and can hold its structure for temperatures up to 300°C. Apart from achieving stability, metal-organic frameworks have also been rendered soft and flexible. They show a dramatic switch in porosity over a short pressure range which significantly changes their adsorption ability.
Based on their molecular structure, metal-organic frameworks can be grouped into different families, such as ZIF, UiO, IRMOF, and HKUST, among others. Scientists have theoretically predicted hundreds of thousands of possible structures of metal-organic frameworks and have synthesized tens of thousands of these. Ongoing research focuses on finding potential applications of these materials and making them more effective.
A world of possibilities

Dr Deepu’s group is working on a range of experimental questions and applications related to metal-organic frameworks.
They are using ZIFs for separating gases like propane and propylene, whose purification traditionally requires more than hundred stages of distillation. Approaches using metal-organic framework membranes offer an energy-efficient alternative for such processes. His group is also studying membrane-based techniques to capture carbon dioxide from industrial emissions which contributes to the United Nations Sustainable Development Goals on clean energy and climate action.
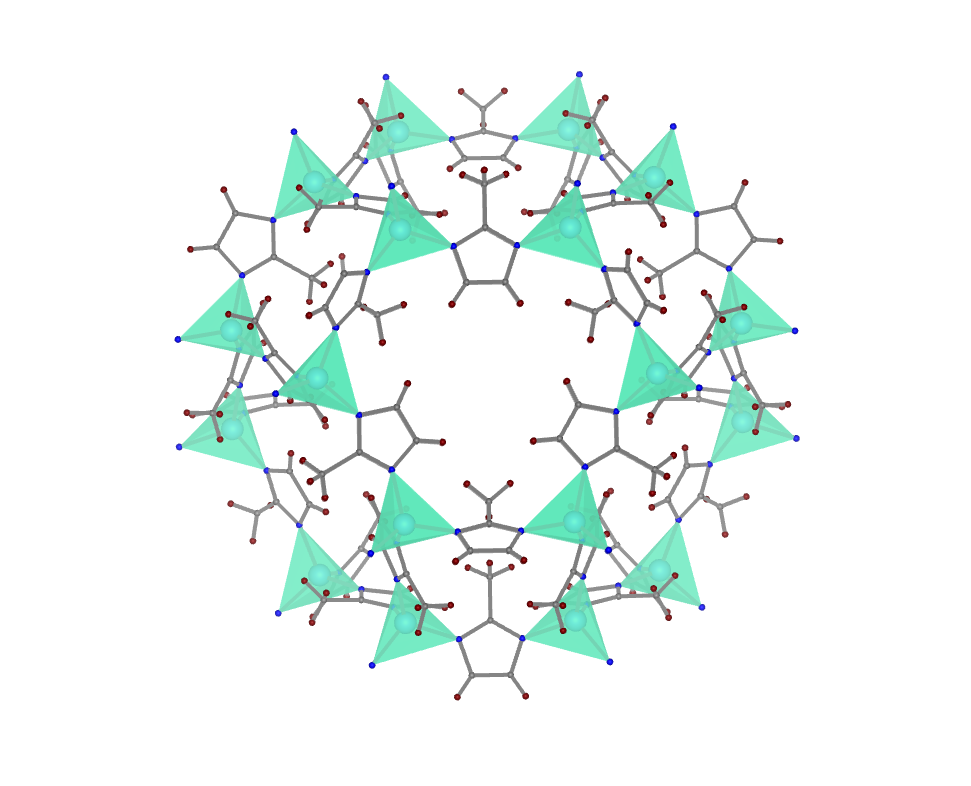
In collaboration with the Defence Research and Development Organisation, Dr Deepu’s group is exploring the use of materials like MOF-801 in extracting water from air in arid regions. “This would help people to get potable water in extreme conditions such as deserts,” he explains.
With metal-organic frameworks, the possibilities seem endless.